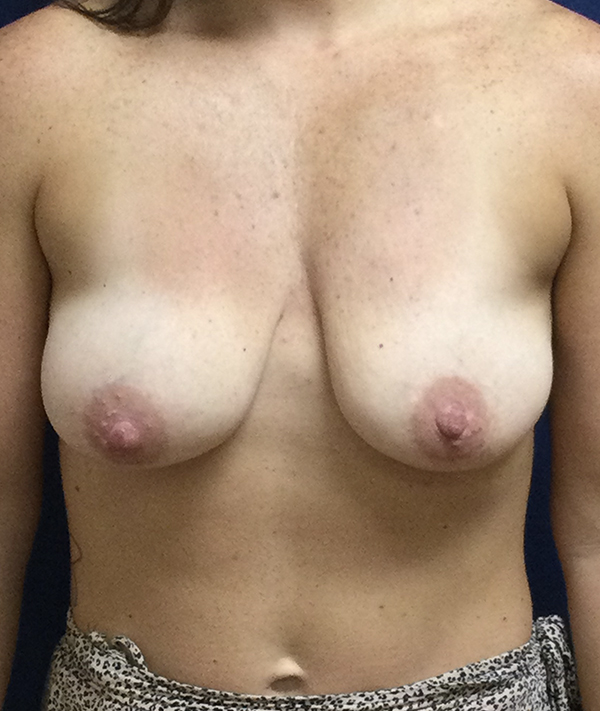
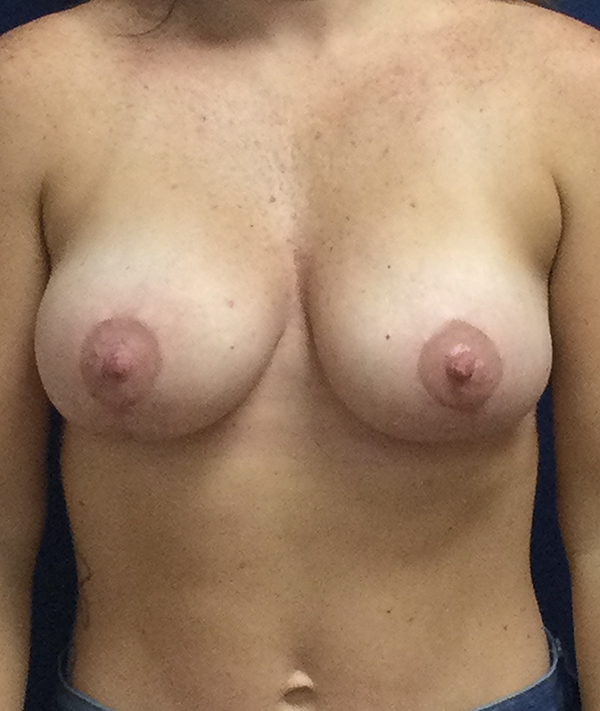
Breast Augmentation Recovery Timeline
Recovery from breast augmentation in Houston is a personal issue. How your body responds to surgery, your natural healing abilities, and other factors unique to your procedure will make your recovery different from anyone else’s. It is reassuring that Houston breast surgeons Dr. Michael Eisemann and Dr. Bradley Eisemann, along with their professional staff, will support you through the entirety of your recovery and are available to you should any concerns arise.
Week one
Mild exercise such as walking should be resumed on the day of your surgical procedure. Frequent walks can help reduce swelling and speed up recovery. You should avoid more strenuous activities that can impact your incisions until Dr. Michael Eisemann or Dr. Bradley Eisemann advise otherwise. Generally, this is within two to six weeks.
Week two
Swelling and bruising may continue for several weeks, but most women are able to resume work and some normal activities within five to seven days. During your initial consultation, we will discuss with you how long you should take off of work based on the physical demands of your job.
Week four
The swelling and bruising are over, and your implants have settled into place, in most cases. Your new breasts are ready to be revealed in low-cut tops, lingerie, or a swimsuit.
Week eight
You should have achieved almost a full recovery. If your implants were placed through the breast fold, the scar is still visible and will usually fade to a thin line over the weeks and months ahead. It is hidden beneath the breast mound, and not easily visible.